CIRS: A New Paradigm for Chronic Disease | Part 4
CIRS Treatment Protocol

Aaron Hartman MD
February 21, 2019

Subscribe
Never miss out on new content from Dr. Hartman.
Interactive Infographic
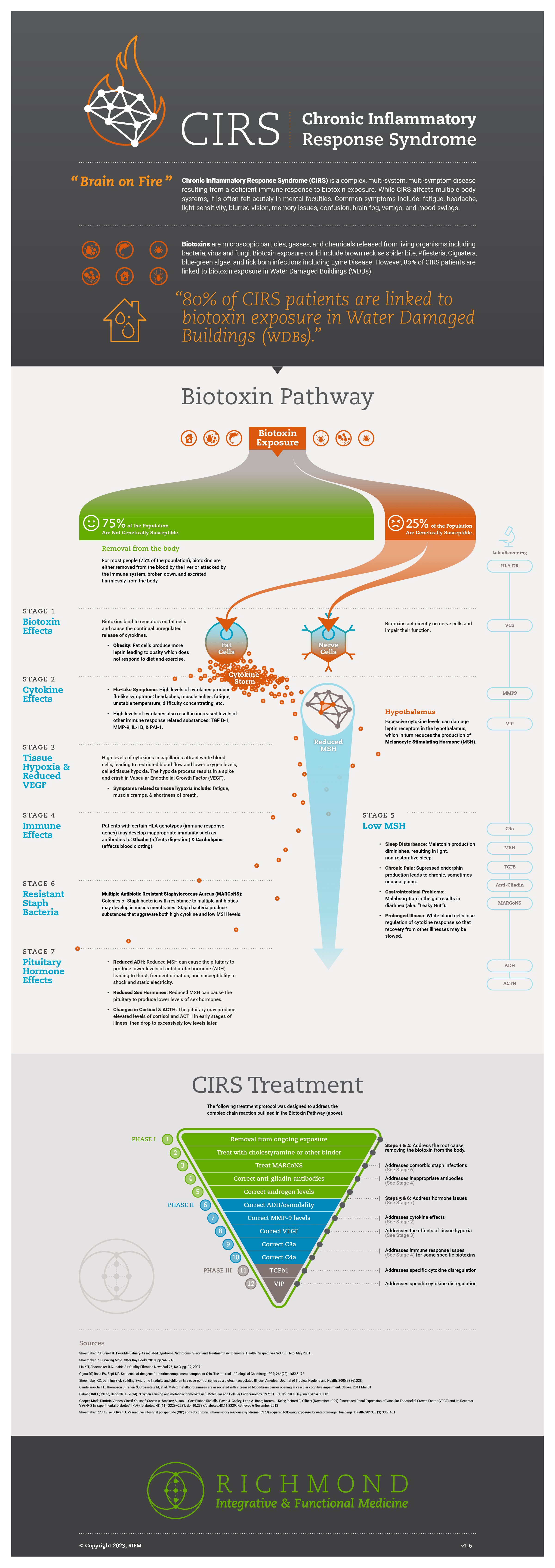
The treatment protocol outlined in this article builds on an understanding of the Biotoxin Pathway — the complex chain reaction when you are exposed to a biotoxin and genetically susceptible to that exposure. If you have not already done so, I encourage you to explore the previous article in this series, or the following infographic that summarizes the process.
Steve’s Story
When I saw at Steve at his one year follow up, the summer of 2018, everything had changed. He had recently gotten a job working as a medical sales representative and moved up north. By his admission, he was working 12-hour days and staying up late during the work week. His energy levels were intact, and he had started to gain back weight (he lost about 30 lbs. of muscle mass from his illness). His thinking was clear, and his memory had come back to him. His only remaining complaint was his new-found sensitivity to any water-damaged buildings. He could tell within 15 minutes of entering a building whether there was any mold toxicity. He would start to get a tremor, ache and feel dizzy.
What steps had Steve taken to regain his health? How had the vigor he once had prior to his illness returned? What lessons from his case could others learn from?
In this article, I will walk you through how to treat CIRS, through the treatment pyramid known as the Shoemaker Protocol.
NOTE: Due to the complexity of CIRS and the treatments involved, patients should only work with a certified practitioner or a medical doctor trained in the Biotoxin Pathway model-of-treatment. The following are only recommendations and should be reviewed with a licensed practitioner.
Part Four:
CIRS Treatment Protocol
A Guide to Treating Biotoxin-Related Illness
The following diagram is a simplified outline of the treatment and protocol for CIRS. The treatment is divided into three phases consisting of a total of 13 steps. The final step (13) consists of retesting the labs and VCS. All of the following steps are initiated only after a definite diagnosis of CIRS has been established by the criteria from Part 2.

Does it matter which way the triangle points?
If you’ve searched the internet or worked with another provider, you’ve probably seen the Stepwise Pyramid pointing up. Does it matter? I am presenting the same twelve-step protocol, but since most of us read from top to bottom, I’ve found it simpler to present the pyramid pointing down.
CIRS Treatment: Step-by-Step
Shoemaker Protocol: Phase 1
1. Removal from ongoing exposure
To remove the ongoing exposure, it must be identified. This is the first and most crucial step of this process. Potential biotoxin exposures include: Lyme, Ciguatera, Pfiesteria and mold. Mold accounts for 80% of biotoxin exposure and many times individuals have multiple exposures (e.g. mold + Lyme). A MSqPCR test commonly referred to as an ERMI is the optimal test for this. “Insider Indoor Air Quality” is an article which explains the deficiencies of current mold spore trapping and the benefits to ERMI testing.
A positive ERMI for someone with CIRS is defined as >2 if their MSH is less than 35. If their C4a testing was >20,000 and MSH<35, then a safe ERMI is <-1. After remediation, once the HERTSMI-2 is <11, the building is safe to enter again.
2. Treat with Cholestyramine or Other Binder
Biotoxins from water damaged building must be removed from the body, this is especially true for those with a genetic predisposition to CIRS. This can be accomplished using cholestyramine (aka CSM), a bile acid sequestrant. The structure of CSM is such that it binds negatively charged ionophores resulting in their excretion in the bile. Biotoxins are ionophores that are difficult to excrete and CSM facilitates their removal from the body. If CSM is not tolerated, other binders can be used but the next best one, Welchol, only works ¼ as well as CSM in binding the ionophore biotoxins. Activated charcoal, sterols and chlorella bind this class of toxins even less potently than Welchol.
There are three types of CSM:
- “Regular” which has 3.5 grams of sugar per dose and some other additive.
- “Light,” which contains aspartame, an artificial sweetener that some patients with Multiple Chemical Sensitives will react with.
3. “Pure resin,” which has no additives.
While the pure resin is more expensive, it is also better due to its lack of additives, though it is significantly more expensive and not covered by insurance since it must be compounded by a compounding pharmacy.
The protocol for dosing cholestyramine is as follows:
- Start with ½ a packet or ½ a scoop daily mixed with 6-8 ounces or water or juice and build up to four times daily over 1-2 weeks. Take CSM an hour before a meal or an hour after a meal.
- If you develop constipation, consider soluble fiber, psyllium, magnesium citrate or high dose buffered vitamin C to maintain at least one bowel movement daily.
- Continue dosing the CMS four times daily for 6 weeks and until the patient passes their VCS testing.
- Common side effects include gas, bloating, reflux, heartburn, nausea and constipation
- Some patients will use Welchol or other binders instead of CSM due to its side effects. These patients tend to be the more chemically sensitive and medically fragile patients.
- If re-exposure to mold occurs, repeat the dosing of CSM at four times daily for three days.
Some patients will develop an acceleration reaction when they first start CSM due to the degree of cytokine elevation in their bodies. These individuals will complain of worsening symptoms and at times will develop new symptoms. This does not mean CSM can’t be used, but further precautions must be taken. These include:
- low amylose diet (see Appendix G)
- EPA/DHA dosed at 3000-4000mg daily for one week then retry the CSM
- consider starting Actos 15-45mg a day as tolerated if the above aren’t’ effective.
Certain medications need to be taken 2 hours after the CSM dosing. These include thyroid hormones, thiazide diuretics and coumadin. Recheck the VCS after one month of CSM treatment and the with each additional step in the treatment protocol. Once the VCS normalizes, the CSM dosing can be cut back to either a once a day preventative, Welchol 625mg twice daily or if the home ERMI was safe and the individual doesn’t go out much, binder treatment may stop. If treatment with CSM fails consider the following: continued exposure, false negative ERMI, wrong diagnosis, non-compliance or untreated MARCoNS.
3. Treat Multiple Antibiotic Resistant Staphylococcus Aureus (aka MARCoNS Treatment)
MARCoNS treatment consists of using an antimicrobial biofilm reducing compounded nasal spray. Traditionally, this was BEG (0.2% Bactroban, 1% EDTA and 0.5% Gentamycin) sprayed twice in each nostril three time daily for 6 weeks. However, early in 2018 this combination was changed to EDTA 1% with colloidal silver 25ppm in Mucolox 15%. Most compounding pharmacies will not make this product, so some investigation may be required to find one. Recent studies have shown this new combination to be effective without the potential ototoxicity of gentamycin. Instructions are the same as with BEG Nasal spray.
After the initially 6 weeks of treatment, discontinue the nasal spray for 5-10 days and reculture for MARCoNS. If negative, then continue with the Biotoxin Pathway treatment. If positive, resume treatment. If the infection is difficult to eradicate, consider re exposure, continued suppressed MSH, partner with suppressed MSH and MARCoNS, dog exposure or non-compliance. If unable to clear after two to three rounds, the practitioner may either revert to BEG Nasal spray for a round of treatment or perform and extended round of EDTA/Silver nasal for three months, then retest.
Do not treat nasal fungal infections with azole antifungals! Research has shown these are transitory and not pathologic. Treatment of these organism found on the API-Staph Culture has been showed to increase the degree of antibiotic resistance in MARCoNS.
4. Correct Anti-Gliadin Antibodies
Anti-Gliadin antibodies were among the first antibody responses noted to be associated with CIRS. Over time others have been documented, but this one was the one initially researched in CIRS when the treatment protocol was being developed. If someone has a positive anti-gliadin antibody, then they need to be 100% gluten free. If an individual is found either on lab testing or endoscopic biopsy to have Celiac Disease, then all gluten containing foods must be removed. Appendix H has a summary of gluten cross reactive foods. If a patient has Celiac Disease but isn’t improving on a gluten fee diet, refer to this chart to determine if they are getting exposed to cross reactive foods.
The no amylose diet of Appendix G is also a no gluten diet. After three months of a no gluten diet, recheck the anti-Gliadin antibodies. If normalized, gluten can be re-introduced as tolerated. If the patient feels well off gluten, then they may choose to remain off gluten for an extended period.
5. Correct Androgens
Ideally, the dysregulated gonadotropins, ACTH, cortisol and testosterone will be corrected as the biotoxin treatment is implemented in a stepwise process. However, this does not always occur. A low MSH will disrupt the typical fluctuations of LH and FSH and the androgens. With an elevated aromatase activity typical in CIRS, just giving testosterone can result in elevated estrogen levels. Part of the protocol is checking a full hormone panel to allow a full view of the steroidogenic pathway. This includes: free and total testosterone, estradiol, estrone, SHBG, LH, FSH, DHEA, DHEA-S and pregnenolone.
If DHEA is still low by the time of Step V, a short-term dosing of DHEA for several months can be considered. DHEA has immune regulatory effects and has been used for autoimmune conditions such as Lupus, the John’s Hopkins Lupus Center currently is doing this.
If the levels of DHEA-S are low, the dosing regimen is as follows: in men 25-75mg daily, in women 5-25mg daily. This is not supraphysiologic, as is the dosing for treatment of autoimmune diseases. Other treats used to raise DHEA include HCG injection and VIP nasal spray. HCG is not part of this treatment regimen and in the Biotoxin Treatment Protocol, VIP treatment occurs later if needed.
Cortisol dysregulation is not treated with hydrocortisone as this suppresses adrenal function. Cortisol dysregulation is addressed by the entire treatment protocol and its effects on the hypothalamic-pituitary-adrenal axis. Please avoid any prednisone or cortisone supplementation unless a physician has property diagnosed Addison’s Disease and is using the corticosteroids for its treatment.
Shoemaker Protocol: Phase 2
6. Correct ADH/osmolality
Antidiuretic hormone is crucial in regulating the bodies blood concentration of electrolytes, known as osmolality. If at this stage in CIRS treatment, there remains a degree of ADH/Osmolality dysregulation, DDAVP support may temporarily be provided. DDAVP is only used in cases of dysregulation where the ADH is absolutely or relatively low related to the osmolality:
- Osmolality is high > 295 or
- Osmolality > 292 and ADH < 2 (relatively low ADH)
After repeat labs verify this continued dysregulation and a normal to high normal sodium level, DDAVP 0.2mg orally is begun every other night for 5 doses. After the 5th dose, blood serum is retested for osmolality and electrolyte levels. If these are both within the normal range and symptoms persist, then the DDAVP is advanced to 0.2mg nightly. Repeat testing in another 5 doses. Some patients will need 0.2mg twice daily, these patients must be watched closely for any aberration in their osmolality or electrolytes. Once the ADH/Osm appears to be regulated, taper slowly off the DDAVP, like how it was started. As a reminder, those with acquired von Willebrand’s disease may need DDAVP as needed for the nose bleeds caused by mold re-exposure.
Correction of ADH/Osm may occur as quickly at 10 days, so be sure to follow the labs on a weekly basis. If any odd symptoms occur while on DDAVP, stop the treatment immediately and recheck the serum osmolality and electrolytes. On a side note, DDAVP can also help to correct and lower C4a levels and MMP-9 levels
7. Correct MMP-9 Levels
Biotoxins can cause cytokines to become elevated resulting in the release of MMP-9 from neutrophils and macrophages. Using CSM to remove the biotoxins starts this process but at times additional therapies are needed.
This cytokine is a marker to tissues inflammation. Matrix Metalloproteinase-9 allow inflammatory mediatory to migrate through tissues in response to cellular mediators. It breaks down the extracellular matrix and helps to propagate an inflammatory response. Elevated levels are seen in CIRS, but it is also elevated in the lungs of asthmatics.
Upregulation of PPAR gamma receptors lowers MMP-9 levels. This can be accomplished by high dose omega 3 fatty acids dosing them at a daily dose of EPA 2400mg and DHA 1800mg. In combination with a low amylose/mold diet (see Appendix F) this is a powerful tool to lower MMP-9. Many times, I will start omega threes at the beginning of the protocol to help patients tolerate CSM better and progress through the protocol quicker.
If levels remain elevated, a specific PPAR gamma agonist known as pioglitazone may be used. This is a diabetes medication that has been in use for over fifteen years. It is used for 30 days along with a low amylose diet after which time pioglitazone is halted but the diet is continued. Typical dosing is 45mg daily. Side effects include low blood sugars, dizziness and nausea. Low term use (i.e. years) has been associated with bladder cancer.
Since pioglitazone also lowers leptin levels, in combination with the diet, it is a powerful weight loss tool. If a person’s leptin level is <7, or age <18, they shouldn’t take pioglitazone. Other effects of pioglitazone include: lowering TNF, PAI-1 and raising VEGF.
Other treatments to lower MMP-9 levels include: Astaxanthin 12mg daily, Resolvins (in SPM Active), butyrate (which increases resolvins), resveratrol 200mg twice daily, berberine, astragalus, scutalleria, curcumin, cordyceps, phosphatidyl choline, glutathione, glutamine, progesterone, pycnogynol and camelia sensensis. But these are not part of the official Biotoxin Pathway treatment protocol.
After thirty days of treatment, recheck labs and proceed in the protocol if indicated.
8. Correct VEGF
Steps 7 and 8 are often done together, as both can lower and elevated VEGF. If still elevated, additional CIRS treatments begin with graded exercise. Graded exercise pushes the cells capacity to utilize oxygen for production of energy, but it must be done without exceeding the anaerobic threshold, if this occurs a patient will get the ‘push/crash phenomena,’ meaning it can take days to recover from a simple exercise regimen or increase in activity levels. An example of a program is the following:
- Begin with a cardio workout for only 5 minutes daily. Increase one minute daily, as tolerated until the program is 15 minutes in duration.
- Once able to tolerate 15 minutes of cardio daily, add on 5 minutes of floor exercises.
- Do the same as above, increase this one minute daily as tolerated until this part of the routine is also 15 minutes daily.
- Add a program of free weight exercises for 5 minutes daily, again increases one minute a day as tolerated until this part of the program is 15 minutes in duration.
- Once tolerating this level of exercise, go back to the previous sections of the 45-minute workout and increase the intensity. Again, as tolerated.
Low dose erythropoietin 8,000IU twice weekly for 5 doses has been used in the past as part of the protocol to lower VEGF. However, due to medical/legal hurdles as well as insurance coverage, it is no longer used. VIP also can help correct VEGF, but it is the last step of the treatment protocol.
9. Correct C3a
Retest levels of C3a and if still elevated, treat. Levels of C3a aren’t often elevated in CIRS-WDB because bacterial cell membranes are required for its elevation. If high, consider Lyme Disease. Does any of the history or HLA DR gene testing suggest this diagnosis? Did the patient have intensification with CSM? Discussing diagnosis and treatment of Lyme Disease is out of the scope of this paper, but some consider Chronic Lyme or Post Lyme Syndrome to be a variant of CIRS. If C3a remains elevated, an ongoing Tick Bite Illness needs to be looked for and treated. If this is not the case, then continue as follows.
The official Biotoxin Treatment Protocol recommends high dose statins to low C3a. These include lovastatin, pravastatin, rosuvastatin, atorvastatin or Fluvastatin. The medication is taken for 30-60 days with Co Q 10 150mg to 300mg.This class of medication does have some serious side effects. For this reason, many patients prefer not to use this class of drugs. If this is the case, there are other alternatives, but they are not part of the official protocol. These include:
- Red Yeast Rice with CoQ10
- Chlorella
- Topical glutathione
- IMD, DMSA or EDTA suppositories
- Detoxification support for heavy metals or persistent organic pollutants
10. Correct C4a
C4a levels are used to assess the overall severity of CIRS. For many, by the time they reach Step X, after removing from exposure and walking through the previous steps, the C4a level has normalized. If not, in the past, Procrit was used at this stage of the protocol to lower C4a. However, due to medical legal issues, insurance non-coverage (medication costs over $4000) and its black box warning due to potential side effects, Procrit is not used as much anymore.
However, VIP used in the last step of the protocol works great for lowering C4a.
Most practitioners now use VIP in Step XIII to lower C4a levels. It works well and has a much better side effect protocol. Lowering the C4a level can improve the cognitive deficits seen in CIRS. These include memory problems, word finding and concentration difficulties, assimilation of new knowledge, confusion and disorientation.
Shoemaker Protocol: Phase 3
11. Lower TGF beta 1
Transforming Growth Factor beta 1 is a cytokine involved in literally transforming tissues. High levels of TGFB1 are associated with hard to treat asthma attacks, fibrosis in organs such as the liver/kidney/lungs and autoimmunity. The level of TGFB1 can be lowered with a blood pressure medication called losartan. This angiotensin receptor blocker has a metabolite EXP 3179 that is associated with its effects in CIRS. Losartan is also used in Marfan’s Syndrome to reduce the risk of aneurism formation, likely due to its antifibrotic actions. Losartan also prevents the conversion of Th17 cells to T reg cell, another mechanism of its actions.
Most patients with TGF beta-1 levels less that 5,000 are not symptomatic and don’t require treatment with losartan. The protocol for losartan use is as follows:
- Begin with 12.5mg once a day and slowly increase to twice daily. If the medication is tolerated, then increase to a maximum dose of 25mg twice daily. Again slowly. In pediatric patients start this medication at 0.6-0.7mg/kg/day in divided doses.
- Monitor blood pressures daily and TGFB1 monthly. Taper off losartan once TGFB1 I less than 5000.
Vasoactive Intestinal Peptide, in Step XIII also lower TGFB1 so anther consideration is moving on to this step if losartan not tolerated, the patient is not willing to take it or there are other contraindications. Other treatments for elevated TGFB1 that are not part of the Biotoxin Treatment Protocol include:
- Resveratrol 200mg daily
- Carnitine 1000mg daily
- Berberine, astragalus, scutelleria, artemisia, Schisandra chinensis, ginkgo biloba, panax ginseng.
12. Vasoactive Intestinal Peptide (VIP)
Vasoactive Intestinal Peptide is the crown jewel of the Biotoxin Pathway treatment. Its actions allow it to lower the pulmonary artery systolic pressure associated with shortness of breath, elevated VEGF, restore the sleep/wake cycle, regulate Th17 function and autoimmunity and even improve the brain volumes seen on NeuroQuant.[1] [2] [3] [4] [5] However, prior to use, all the following criteria must be met:
- Patient is in a safe environment as defined by MSqPCR testing (ERMI<2 or HERTSMI 2<11)
- VCS normalized
- MARCoNS eradicated
- Already have walked through the previous eleven steps of the Biotoxin Protocol
- Lipase level is normal
Another consideration is a stress echocardiogram to establish pulmonary arterial pressure. A high PASP (>30mmHg) at rest or an increase of over 8mmHg in response to exercise are also criteria for VIP use, though not a part of the ‘official’ protocol. This elevation is what is translated into the symptoms of breathlessness, shortness of breath or air hunger or fatigue/shortness of breath with slight exertion. The PASP is calculated by the following formula:
PASP=RA + 4(TR2)
Pre-administration labs should be drawn and include the following: VIP, MSH, TGFB1, C4a, VEGF, MMP-9, Lipase, comprehensive metabolic panel, Vitamin D, Estradiol and total Testosterone. The first dose is administered in the office for safety precautions.
The following is the VIP protocol:
- Give one spray of VIP into the nostril of the patient. After 15 minutes draw the C4a and TGFB1 levels. If there is a two-fold increase in these labs, it can be a marker for continued mold exposure.
- Vital signs are checked every 5 minutes over 15 minutes. The provider looks for any symptoms or reactions but especially development off a rash, abdominal pain, blood pressure
- Fluctuations reduced joint pain, improved mentation and improved shortness of breath.
- If the patient has no adverse side effects to the trial and the TGFB1 and C4a done douuble, then VIP can be begun at 50mcg (1 spray) intranasally four times a day for one month. After that time the dose is dropped back to twice a day for one month then daily for one month. The lipase level is checked monthly if a patient is on VIP.
NOTE: Pancreatitis is a potentially dangerous reaction to VIP. If while on therapy the patient gets abdominal pain, a rash or an elevated lipase level, VIP must be stopped immediately.
Before starting VIP therapy, most will have already had an 75% reduction in their symptoms. VIP can help patients to regain even more function and symptom reduction. Patience is key. It can take anywhere from nine months to two years to go through the above process. It is grueling work and costly at times, but for some individuals, it is the only way to improve their health and gain their life back.
Conclusion
This entire process will require a re-orientation of an individual’s thoughts, actions and lifestyle. Most will not even start the process and of those who do, even fewer will complete it. But for individuals with CIRS this may be the only way to regain their health. What I have put together in these four parts is meant mainly as a primer. It is no way replaces working with a skilled licensed experienced clinician who has worked with difficult cases in the past.
Other Resources
The following are additional resources that can help both the patient and professionals that are working with them. Please make use of these. The more education the better. The more you understand what is going on in your body and what others have gone through, the better equipped you will be to make this journey.
This kind of medicine requires the patient and family to be active participants in their care. You will need to be an active participant in your care. At the same time, there are many resources out there that are subpar and only superficial in their approach and understanding of CIRS. Be aware of these as they result in delayed treatment and worsening of the underlying medical condition. Please review all the resources below. These will enable you to become engaged in your health care and empowered in the struggle it will be to work through this.
-
Mold Illness Surviving and Thriving: A Recovery Manual for Patients & Families Impacted By CIRS
This is a patient clinical manual put together by a group who has been working with patients for years. It is meant to be used by a patient while they work with a practitioner to help guide them through this long and complicated process. The book could be compared to a longer version of this paper but written for the express purpose to guide patients through their treatment. It is a must-read for those either working through their own illness or the loved ones helping guide them through it.
-
Chronic Inflammatory Response Syndrome: Consensus Statement
As of the writing of this paper, this is the most up to date physician consensus statement on CIRS, it’s pathology and the basic science behind the disease. This is a must-read for clinicians dealing with CIRS patients. Patients interested in the technical data should also consider reading this.
-
Consensus Statement on CIRS by Indoor Environmental Professionals 2016
This paper reviews the science behind what was once known as ‘Sick Building Syndrome.’ How is it that a building can make a person sick? How does one start the remediation process? This paper reviews the exposure side of CIRS reviewing the date and literature behind water-damaged buildings and how they affect our health. This is a must-read for mold inspectors, building inspectors and homeowners.
-
Surviving Mold: Life in the Era of Dangerous Buildings by Ritchie C. Shoemaker, MD
This book is written by the physician who initially discovered CIRS in the late 1990’s. It is a history of his struggle to learn how to treat it, the bureaucracy involved in preventing its discovery and treatment, and reviews his initial protocols on treatment. It is a nice review of the history of CIRS and written in a clear and understandable style. The protocols for treatment are constantly changed due to new discoveries, the most up to date treatment protocols can be found under the physician essays found here.
References
- Shoemaker RC, House D, Ryan J. Vasoactive intestinal polypeptide (VIP) corrects chronic inflammatory response syndrome (CIRS) acquired following exposure to water-damaged buildings. Health. 2013;5:396-401.
- Gupta V and Ahsan F. (2010) Inhalational therapy for pulmonary arterial hypertension: current status and future prospects. Critical Reviews in Therapeutic Drug Carrier Systems. 27:313-370.
- Yu XJ, Ren XH, Xu YH, Chen LM, Zhou CL and Li CY. Vasoactive intestinal peptide induces vascular endothelial growth factor production in human HaCaT keratinocytes via MAPK pathway. Neuropeptides. 2010;44:407-411.
- Gonzalez-Rey E and Delgado M. Role of vasoactive intestinal peptide in inflammation and autoimmunity. Current Opinion in Investigational Drugs. 2005;6: 1116- 1123.
- Shoemaker RC, Katz D, Ackerly M, et al. Intranasal VIP safely restores volume to multiple grey matter nuclei in patients with CIRS. Internal Medicine Review. 2017;3(4):1-14.
Take Back The Outdoors
Get the relief you need:
- Blocks 96% of histamine release BEFORE symptoms start
- Works within 14 days (critical for beating peak pollen season)
- Includes the exact formulation that delivered results in clinical studies
- Has helped 12,549 allergy sufferers enjoy spring without medications
Don’t wait until you’re already sneezing. The prevention window is closing.
10% discount through Fullscript. First-time buyers save an additional 15%.